Medical device risk management template5/25/2023 The RMP should also include the collection process of information from the production phase and from the post-market surveillance system and its impact should be evaluated. The known and foreseeable hazards associated with the device and with the intended use should be identified and analysed, and mitigation strategies should be discussed. Data from the post-production phase needs to be collected and implemented in order to maintain corrective action and vigilance procedures in a systematic way.Īs a first step, a comprehensive Risk Management Plan (RMP) should be compiled, defining the activities associated with risk recognition, mitigation and surveillance throughout the life cycle of the product. Thereafter, the risk management report summarizes the results of all risk management activities, and it should provide justified statement that the benefits outweigh the risks. 
Risk control demonstrates the mitigation measures that are proposed and their implementation where necessary. 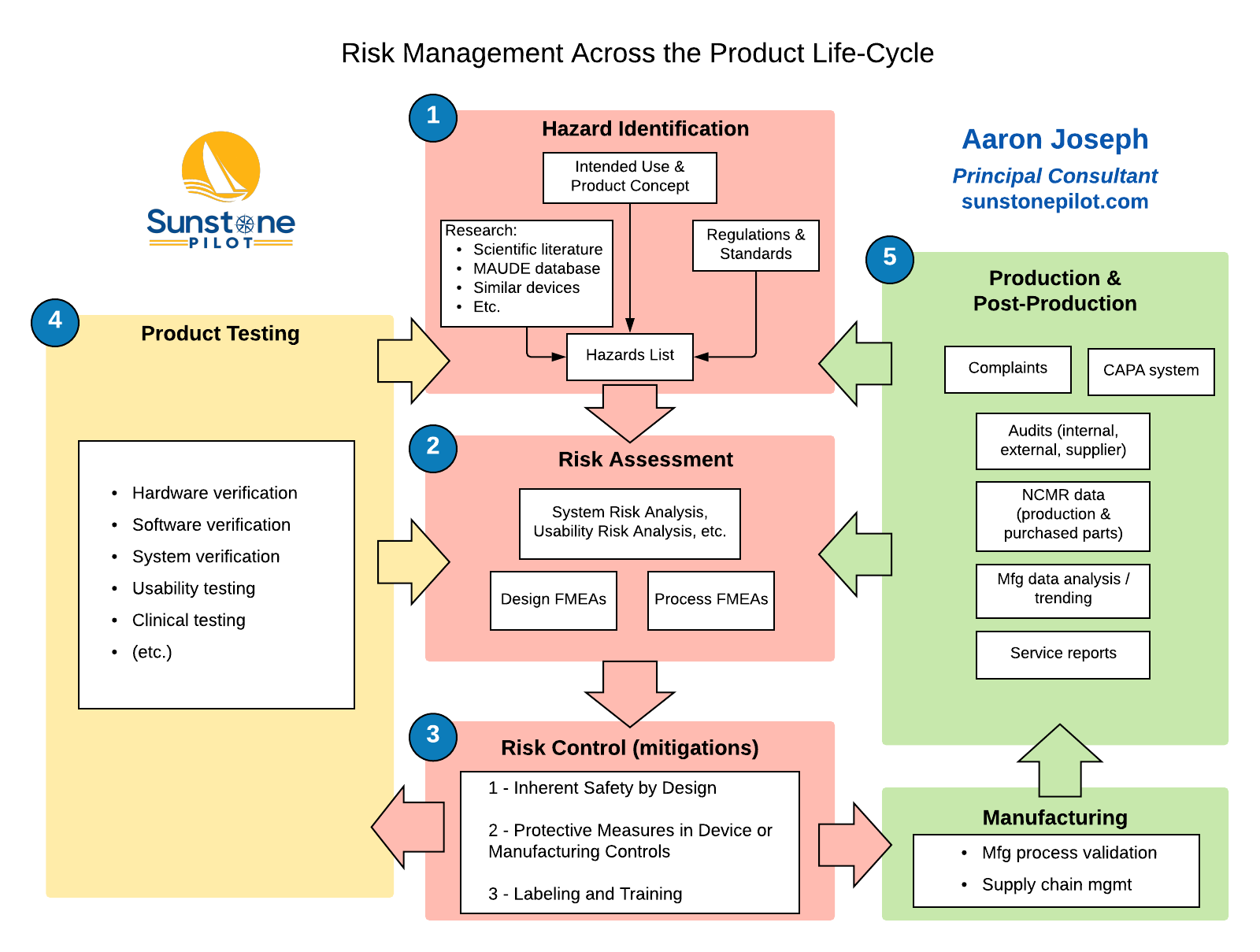
Risk analysis and evaluation include the preliminary hazard identification, associated risk assessments and categorization of the recognized risks. Figure 1: The risk management process: The risk management plan defines the activities associated with risk recognition, mitigation and surveillance throughout the life cycle of the product.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
